 Electrons are transferred from one element to another. The redox reaction is a chemical reaction that produces a change in the oxidation states of the atoms involved. In an electrochemical cell, spontaneous redox reactions take place in two electrodes separated by an electrolyte, which is an ionic conductive and electrically insulated substance. Chemical energy can be stored, for example, in Zn or Li, which are high-energy metals because they are not stabilized by d-electron bonding, unlike transition metals.Įven though a wide range of types of batteries exists with different combinations of materials, all of them use the same principle of the oxidation-reduction reaction. If we go into detail, batteries convert chemical energy directly to electrical energy. This battery chemical reaction, this flow of electrons through the wire, is electricity. zinc) through the wire in the circuit to the cathode electrode. manganese dioxide in alkaline batteries) will pull the electrons from the anode (e.g. When the circuit is closed, the stronger attraction for the electrons by the cathode (e.g. This happens when the battery is placed in a device, and the device is turned on. The stored electrons will only flow when the circuit is closed. In simple terms, each battery is designed to keep the cathode and anode separated to prevent a reaction. Alkaline batteries are most commonly used in portable devices that have low current drains, are used only intermittently, or are used well away from an alternative power source, such as in alarm and communication circuits where other electric power is only intermittently available. 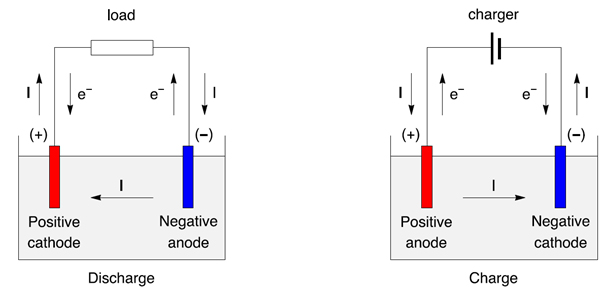
The most common size of alkaline battery is the well-known AA battery. The primary alkaline battery is a widely used product, which is essential for powering many portable devices, such as power tools, radios, toys, and remote controls. Other battery systems also use alkaline electrolytes, but they use different active materials for the electrodes. The alkaline battery gets its name because it has an alkaline electrolyte of potassium hydroxide (KOH) instead of the acidic ammonium chloride (NH 4Cl) or zinc chloride (ZnCl 2) electrolyte of the zinc–carbon batteries. An alkaline battery (IEC code: L) is a type of primary battery that provides direct electric current from the electrochemical reaction between zinc and manganese dioxide (MnO 2) in the presence of an alkaline electrolyte. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
